The World Health Organization greenlighted emergency use of AstraZeneca and Oxford’s covid-19 vaccine this month, following in the steps of the United Kingdom, the European Union and others, who are already injecting it as quickly as possible into the masses.
But the United States is still waiting.
As covid deaths mount daily, critics say the Food and Drug Administration is moving too slowly. Meanwhile, the novel coronavirus is evolving, with new variants stalking populations the world over.
“We are truly in a race and this race is real — the more we get people vaccinated, the more it will tamp down the virus that is mutating,” said Dr. Monica Gandhi, an infectious diseases specialist and professor of medicine at the University of California-San Francisco.
The world has seven vaccines with completed clinical trials, yet the U.S. has approved only two, Gandhi and others lament.
AstraZeneca — after global trials that included some mistaken dosing — has not filed an application in the U.S., saying it first needs to finish its phase 3 U.S. trial. Simply put: AstraZeneca hasn’t applied for the U.S. job.
The company knows that the FDA doesn’t merely accept results from trials in other countries. And its confusing trial results pooled from differently designed clinical trials in Brazil and the U.K. raised questions about dosing as well as how well it works for people 65 and older. Germany and France have said not to administer the vaccine to older residents, while the World Health Organization said it was fine to do so.
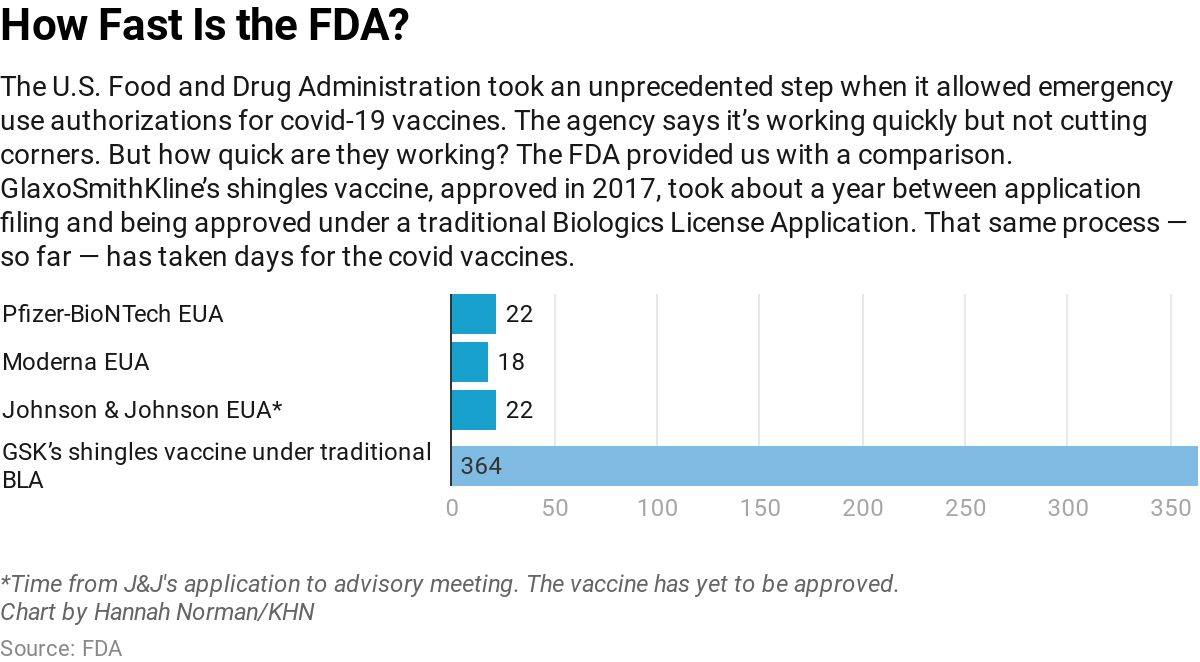
The FDA — one of the oldest drug approval agencies on the globe — issued emergency use authorizations late last year for two vaccines manufactured by Moderna and Pfizer-BioNTech. It is reviewing an application from Johnson & Johnson, which filed Feb. 3, and the advisory panel is scheduled to discuss it at a Feb. 26 meeting.
“The FDA is not the villain here,” said Dr. Cody Meissner, a pediatric infectious diseases specialist at Tufts University who sits on the FDA’s vaccine advisory panel.
Drug approvals usually take months once an application is filed, but the FDA’s emergency authorizations for covid vaccines have been granted within weeks. FDA spokesperson Abigail Capobianco said its staff is working nights, weekends and holidays to prepare for the meeting — moving with a sense of urgency.
“FDA staff are mothers, fathers, grandparents, daughters, sons, sisters, brothers and more,” Capobianco said. “They and their families are also directly impacted by the work that they do.”
J&J’s vaccine, which received in development funds through Operation Warp Speed, uses an adenovirus — a vector that produces cold-like symptoms — to deliver a piece of genetic code that triggers an immune response in the body. It would be authorized in the U.S. — a possible game changer in getting more Americans vaccinated.
“People have been clamoring for it to be approved and everybody wants it to go faster,” said Dr. Amesh Adalja, a senior scholar at Johns Hopkins Center for Health Security who has called for more harmonization between approvals from the U.S., U.K. and European Medicines Agency.
“The question would be from a policy standpoint,” Adalja said. “Would the FDA be willing to say that what the EMA does is equivalent to them and they would have full confidence in the EMA decision?”
Despite the need for speed, the FDA said it will not cut corners. Before last fall, vaccines typically went through a full licensing process before being distributed to the public. The use of emergency authorization to give a vaccine to millions of otherwise healthy people has “never been done” before, said Norman Baylor, a former director of the FDA’s vaccine research and review office who now consults with pharmaceutical companies.
To prepare for J&J’s advisory committee, FDA staff members as well as the independent advisory panel will have analyzed thousands of data points to consider whether the benefit of a vaccine outweighs the risk of injecting it into millions of otherwise healthy people. The FDA is not required to follow the panel’s recommendation but usually does.
Meissner, who abstained in the vote for the Pfizer-BioNTech vaccine, said, “We want every vaccine to succeed.” Everyone on the planet needs immunization — billions of people.
“The more manufacturers that can provide vaccines, the better,” Meissner said. “I don’t think anyone would be against additional manufacturers.”
This moment — as Americans question why more tested vaccines like AstraZeneca and J&J’s vaccines aren’t approved — punctuates how the FDA’s drug approval process, honed over decades, is independent of other global agencies. Dr. Henry Miller, a senior fellow at the Pacific Research Institute who was the founding director of the FDA’s office of biotechnology, said it’s difficult to compare international vaccine development.
“It’s not like a footrace where everyone begins together,” he said. “From country to country, there are a lot of variables.”
Some are trivial, such as different application processes and whether the companies completed the forms properly. Others are more substantial — while many countries depend on academics on contract, the U.S. relies on full-time staffers who spend their careers focused on drug development, Miller said.
Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said that the “FDA gets involved very early on in the process, that makes us unique among global agencies.”
FDA staff members have had discussions with some vaccine makers “about how they would do the work” even before the vaccines went to early clinical trials in humans. They are in contact through the various stages of manufacturing, Marks said.
Technically, AstraZeneca and the other vaccine makers have filed what are called “investigational new product” applications with the FDA. That means the companies early on submit the details of drug formulation, stability and laboratory work. They also provide results data at the end of each clinical trial phase.
AstraZeneca, which was awarded through Operation Warp Speed to develop a vaccine, “remains in close, regular communication” with federal agencies, said AstraZeneca spokesperson Brendan McEvoy.
There are differences in what each country needs from the vaccines. The AstraZeneca vaccine will be “for a very different population than the Pfizer and Moderna vaccines,” former FDA staffer Miller said. The Pfizer and Moderna vaccines are more costly and demand cold-storage infrastructure that many developing countries can’t afford.
Plus, Miller said he believes the international agencies were eager to approve AstraZeneca. “Circumstances suggest they are willing to accept a somewhat lower standard — much like a drug intended to cure cancer makes you willing to accept greater side effects because the need is so great and the benefit is so great. It’s all risk, benefit and probability,” he said.
AstraZeneca’s acceptance abroad is enough for some people. “Why wait for another clinical trial to be completed?” asked Dr. Martin Makary, a professor of surgery and health policy at the Johns Hopkins University School of Medicine in Baltimore. “You have the real-world observation of the vaccine being given to millions of people.”
Gandhi, who has followed the clinical trials from across the globe, expressed more urgency. “The U.K. will get to herd immunity faster,” she said. “All of these wonderful things the FDA is doing that we are all so impressed by are taking too long.”
In October, the FDA released that seek approval in the U.S. It’s “pretty clear what designs were needed in the studies and what the FDA’s expectations were for the data,” said Dr. Jesse Goodman, former director of the FDA’s Center for Biologics Evaluation and Research, which regulates vaccines. He was also the FDA’s chief scientist from 2009 to 2014, leading its response to the 2009 H1N1 pandemic.
The agency asked for evidence that the vaccine’s benefits outweigh its risks based on data from at least one well-designed phase 3 clinical trial. To pass muster, it will need to prevent disease or decrease the severity of the disease in at least 50% of people vaccinated. Both Moderna and the Pfizer-BioNTech vaccines are well above that threshold, at and respectively.
Dr. Stanley Plotkin, a scientist and vaccine developer, said Pfizer and Moderna’s vaccines were greenlighted after large U.S. trials with “very clear results, high efficacy.” One challenge for AstraZeneca will be the variation in data — different trials with different dosages and population numbers. Clinical trials found the vaccine had an efficacy of when two doses were given 12 weeks apart.
The FDA will dig into any incoming research numbers to determine how well each vaccine works with different doses and schedules. They will question whether they prevent serious or mild disease, while accounting for varying age groups of the trial populations, including subsets that may be more likely to get sick. Other aspects up for analysis will be the immunogenicity, or antibody response, and the safety data.
“Asking questions and asking for more data, that is exactly what they are supposed to do,” said Plotkin, now a professor emeritus at the University of Pennsylvania who consults for Moderna and others.
And, since multiple vaccine investigations are underway, FDA staff members will have reviewed the data from various applications — and may have questions that are not obvious to company researchers working on individual projects, said former vaccine regulator Goodman, who is now a Georgetown University professor.
FDA staffs work beyond the numbers as well and often do a “thorough investigation and validation of the plant” where vaccines will be produced, said Kevin Gilligan, a virologist and former unit chief at the federal government’s Biomedical Advanced Research and Development Authority. “You want to make sure there aren’t any remaining pathogens in there and all the equipment used is thoroughly clean and reevaluated,” Gilligan said.
Novavax, which received $1.6 billion through Operation Warp Speed in July, is developing a two-shot protein-based vaccine. After addressing , Novavax ramped up full-scale manufacturing operations. Novavax spokesperson Silvia Taylor said the company has been in “ongoing contact” with the FDA and is “already beginning to submit” various parts of its application and data to agency officials. It expects initial results of its U.S. phase 3 trial before summer.
Taylor said Novavax has already “locked” its manufacturing process at scale and will be ready to distribute in the U.S. as soon as emergency use is approved.
California Healthline editor Arthur Allen contributed to this report.
Â鶹ŮÓĹ Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at Â鶹ŮÓĹ—an independent source of health policy research, polling, and journalism. Learn more about .